This medication is used to treat severe cystic acne (also known as nodular acne) that has not responded to other treatment (e.g., benzoyl peroxide or clindamycin applied to the skin or tetracycline or minocycline taken by mouth). It belongs to a class of drugs known as retinoids. It works by decreasing facial oil (sebum) production. High amounts of sebum can lead to severe acne...
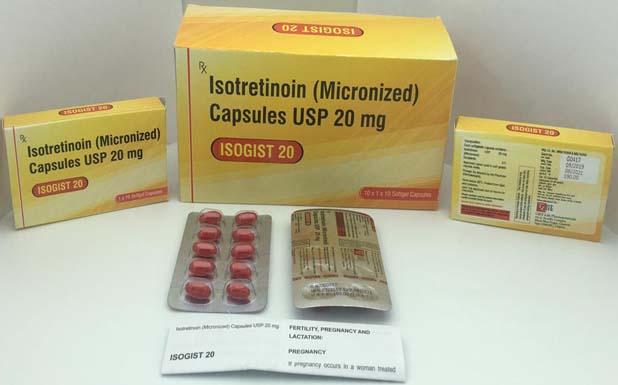
Isotretinoin 5/10/20/30/40 mg
Soft gel
For Derma Care
Severe forms of acne (such as nodular or acne at risk of permanent scarring) resistant to adequate courses of standard therapy with systemic antibacterial and topical therapy.
Isotretinoin is an orally administered systemic retinoid. At a pharmacologic strength of 0.5 to 1.0 mg/kg per day, isotretinoin is an effective acne therapy. While the exact mechanism of action is unknown, isotretinoin inhibits sebaceous gland function and keratinization at pharmacologic doses.
Contraindicated in patients who are hypersensitive to this medication or to any of its components. Should not be given to patients who are sensitive to parabens, which are used as preservatives in the gelatin capsule, in hepatic and renal impairment and hyperlipidemia. Pregnancy must be excluded before therapy and contraceptive measures taken in women of child-bearing potential.
Dry lips, cheilitis (sore, cracked or scaly lips), Dry skin, fragile skin, eczema/dermatitis (itchy, red patches of skin), and Increased sweating.